Ammonium acetate nonetheless remains a useful additive for native ESI-MS. 
This implies that neutral ammonium acetate solutions electrosprayed in positive ion mode will likely undergo acidification down to pH 4.75 ± 1 in the ESI plume. Ammonium acetate does provide buffering around pH 4.75 (the pKa of acetic acid) and around pH 9.25 (the pKa of ammonium). Dissolution of ammonium acetate salt in water results in pH 7, but this pH is highly labile. NH4 + and CH3-COO- are not a conjugate acid/base pair, which means that they do not constitute a buffer at pH 7. The buffering range covers the weak acid pKa ± 1 pH unit. By definition, a buffer consists of a weak acid and its conjugate weak base. It is a widely perpetuated misconception that ammonium acetate buffers the analyte solution at neutral pH. Most native ESI-MS studies employ neutral aqueous ammonium acetate solutions. One potential pitfall is the occurrence of pH-induced changes that can affect the analyte while it is still surrounded by solvent. 
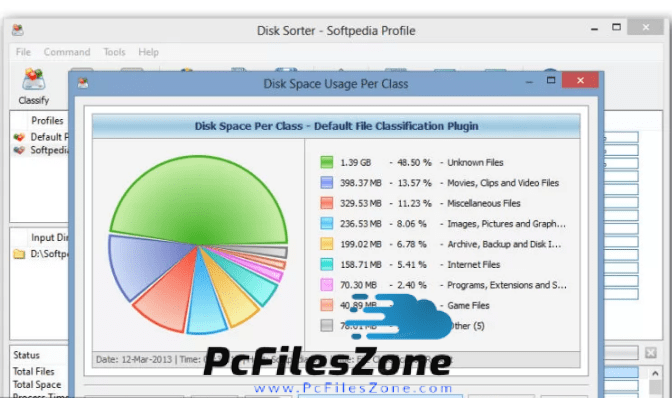
Native ESI-MS involves the transfer of intact proteins and biomolecular complexes from solution into the gas phase. The present invention relates to a composition comprising : a) 0.01-20% wt/wt acetic acid and b) a physiologically tolerable buffer capable of maintaining acetic acid at a pH in the range of 2-7 and use of such a composition as an antimicrobial agent.The present invention relates to a composition comprising : a) 0.01-20% wt/wt acetic acid and b) a physiologically tolerable buffer capable of maintaining acetic acid at a pH in the range of 2-7 and use of such a composition as an antimicrobial agent.Īddressing a Common Misconception: Ammonium Acetate as Neutral pH " Buffer" for Native Electrospray Mass Spectrometry Top Freeware Keywords Show more Show lessĪlSys: Need File Search Portable 1.0 freeware downloadĪllows you to easily search for any files in your system, and allows you to quickly set search wildcards for some popular file formats such as graphic, video, audio, archive or. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
